Healthy Living Articles
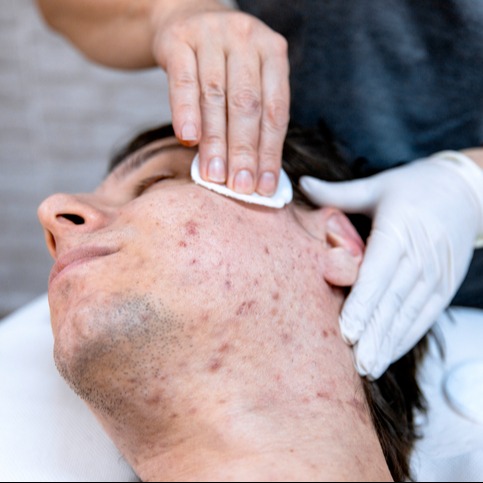
Tretinoin or Retinol? What’s the Difference?
Mar 20, 2024
When I discuss acne treatments with patients, many of them ask me about vitamin A-based medications like tretinoin and retinol. I enjoy these discussions because it indicates that patients have taken the time to research their options and have a baseline understanding. An educated patient is the best patient! Remember to consult your healthcare...
Read More
Clinical Success: Topical Tretinoin for Treating Adolescent Acne Vulgaris
Mar 17, 2024
Author’s Note: The details about “Jamie’s” case are fictionalized to protect his privacy. However, the story of Jamie’s victory over acne is entirely accurate. A licensed healthcare professional should direct any course of treatment with a prescription medicine.Acne vulgaris, commonly referred to as “acne”...
Read More
Colorado Seeking To Join Florida In Importing Prescription Drugs From Canada
Mar 13, 2024
As prescription drug costs have risen over the years, so have concerns related to the impact. These prices aren’t just affecting bank accounts, they’re affecting health, as many individuals are simply unable to take critical medications they have been prescribed. Various solutions have been proposed, and buying prescription medications...
Read More
Flovent Inhaler Availability
Mar 12, 2024
Flovent HFA, or fluticasone propionate, has been a critical component of asthma care for decades. However, the product's availability in the United States has become problematic, and many are searching for solutions to continue managing their breathing symptoms effectively. Flovent DiscontinuedGlaxoSmithKline (GSK) has announced that it...
Read More
Is Artificial Intelligence the Solution to the Physician Burnout Problem?
Mar 7, 2024
I am a victim of physician burnout.The pressures of maintaining a solo private practice wore me down to the point of mental and physical breakdown. I stayed in the profession much longer than I should have. I needed to step away for the sake of my health and the stability of my family.Physician burnout is a critical issue in healthcare, with recent...
Read More
FDA Reminds Older Adults to Be Careful with Medications
Mar 5, 2024
In February 2024, the US Food and Drug Administration (FDA) reminded older adults about safety when taking prescription medications. I am pleased that the FDA issues these reminders, but I wish they had reminded older adults that they also need to be careful with over-the-counter (OTC) medications. When I counsel patients in the office, I remind...
Read More
Outrageous Pricing of Viibryd Sparks Controversy
Mar 3, 2024
he exorbitant pricing of Viibryd, a medication used to treat major depressive disorder (MDD), has garnered significant attention and sparked a contentious debate within the pharmaceutical industry. This controversy highlights the pricing disparities within the United States healthcare system and underscores the lack of regulatory oversight in...
Read More
The Hair-Loss Drug Finasteride Also Reduces Cholesterol
Feb 28, 2024
A remarkable group of prescription medications has gained fame for their ability to treat at least two seemingly unrelated medical conditions. Perhaps the most notable example in 2023 was the finding that the diabetes drug Ozempic also led to weight loss in patients. The somewhat complex regulations surrounding pharmaceuticals often lead to the...
Read More
How Much Is Cialis® (tadalafil): Unveiling the Surprising Cost of Cialis
Feb 20, 2024
In the realm of men's health, one medication has become synonymous with the treatment of erectile dysfunction: Cialis® (tadalafil). However, many individuals may not realize the surprising cost of this widely recognized pharmaceutical solution. As we delve into the factors that contribute to the pricing of Cialis, it becomes clear that the...
Read More


